Publications
Electrochemical Regeneration of Caustic Absorbent for the Capture of CO2
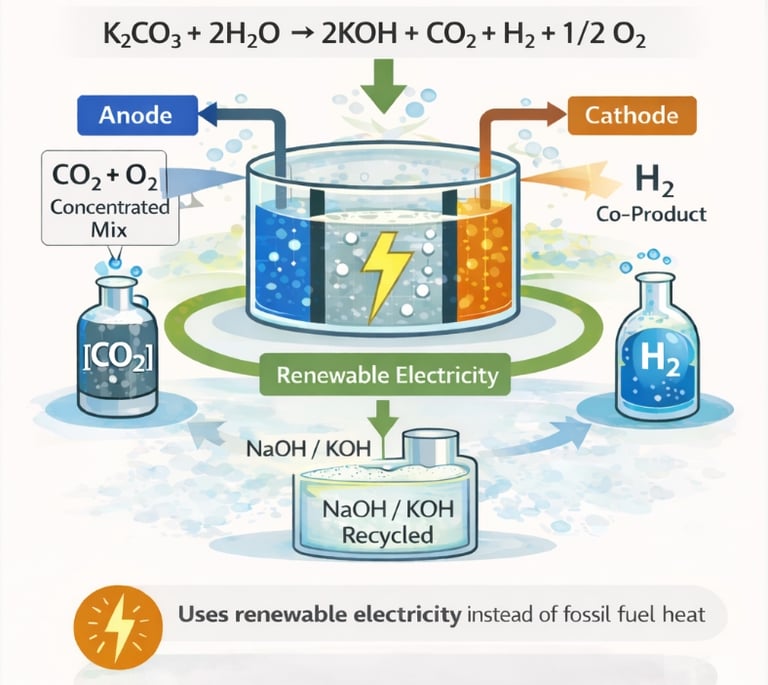
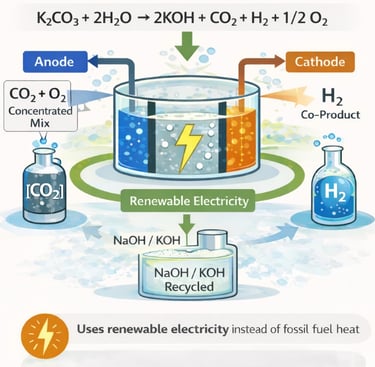
This 2025 study tests a new electrochemical reactor designed to regenerate alkaline solutions (NaOH or KOH) used in carbon capture. Instead of using high-temperature, fossil-fuel-driven heat to convert carbonates back into hydroxides, the system uses electricity to split alkali carbonates into hydroxide and a concentrated mix of CO₂ and oxygen, with hydrogen as a co-product. The reactor avoids costly ion-exchange membranes and operates at relatively high current densities, demonstrating technical feasibility at the lab scale, though efficiency drops over time and materials degrade under harsh conditions.
The publication is important because it explores how carbon capture—especially direct air capture—could be powered directly by renewable electricity rather than heat from fossil fuels. By simplifying reactor design and addressing scalability challenges, the study advances electrochemically mediated carbon capture as a potential low-carbon alternative for CO₂ removal. While not yet commercially viable, it represents a meaningful step toward more sustainable, electricity-driven carbon capture systems aligned with net-zero climate goals.
Development and use of a Mixed-Reactant Fuel Cell
This publication research focuses on mixed-reactant fuel cells (MRFCs), an innovative type of electrochemical power source. These fuel cells combine fuel and oxidant into a single unstable mixture to generate electricity efficiently. The study developed and tested these fuel cells using a potassium formate (liquid) and oxygen (gas) mixture as the fuel and oxidant. Key achievements include:
Scaling from a single cell to a 19-cell stack.
Achieving high power density and efficiency with palladium/silver catalysts under specific operating conditions.
Successfully using these fuel cells to power a modified electric scooter, marking the first known application of MRFCs as the sole power source for a vehicle.


Electrochemical Processing of Carbon Dioxide
Given the impact of carbon dioxide (CO₂) on climate change, there is a strong need for processes that turn CO₂ into valuable products. One promising approach is electrochemical reduction, which can produce useful chemicals like formic acid, methanol, or methane under mild conditions. Recent studies show that CO₂ can be converted into formate (a chemical used in various industries) using a continuous electrochemical reactor operating efficiently at a laboratory scale.
While challenges remain, such as cathode stability and preventing product loss, this technology could be scaled up to convert large amounts of CO₂ (around 100 tonnes per day) into commercial products like formate salts or formic acid, offering a practical solution for reducing emissions and creating useful materials.
Surfactant-Enhanced Oxygen Electroreduction to Hydrogen Peroxide
This study focuses on an electrochemical process to efficiently convert specific chemicals under controlled conditions, using specialized materials like graphite felt or carbon-based electrodes. The research highlights the use of a surfactant (Aliquat® 336), which improves the efficiency of the process by preventing unwanted reactions and reducing energy losses. Although the surfactant increases pressure in the system, it significantly boosts performance, making the process more effective.
As an environmental solution, this method offers potential applications in cleaner chemical production and carbon recycling technologies. By optimizing electrochemical reactors to operate efficiently, it contributes to sustainable energy processes and reduces harmful emissions, supporting a move toward greener industrial practices.
Direct Methanol Fuel Cell with Extended Reaction Zone Anode
The direct methanol fuel cell (DMFC) holds great promise as a clean energy solution for electronics and transportation, but challenges like fuel inefficiency and CO₂ buildup have limited its progress.
A breakthrough anode design, featuring a thicker reaction zone and fibrous structure, efficiently tackles these issues—boosting performance and reducing energy loss. By innovatively coating graphite felt with platinum-ruthenium nanoparticles, researchers have unlocked new possibilities for cleaner, more efficient fuel cells. This work inspires hope for sustainable energy advancements, bringing us closer to a future powered by smarter, greener technologies.




Extended Publication List
Explore Colin Oloman's list of 80 publications on ORCID.
Colin's contributing ID is 0000-0002-7423-8332.